
On consecutive days, each participant will be randomly assigned to receive either the FDA-approved Pfizer COVID-19 mRNA vaccine, Comirnaty, or a look-alike dose of inactive placebo. To maximize safety, all participants will be admitted for a minimum of four days to the Intensive Care Unit at the NIH Clinical Center, where experienced medical staff, as well as equipment and medications needed to treat severe allergic reactions, are readily available. “Our study aims to provide a better understanding of the mechanisms responsible for systemic allergic reactions such as hives, swelling, trouble breathing and light-headedness or passing out.” 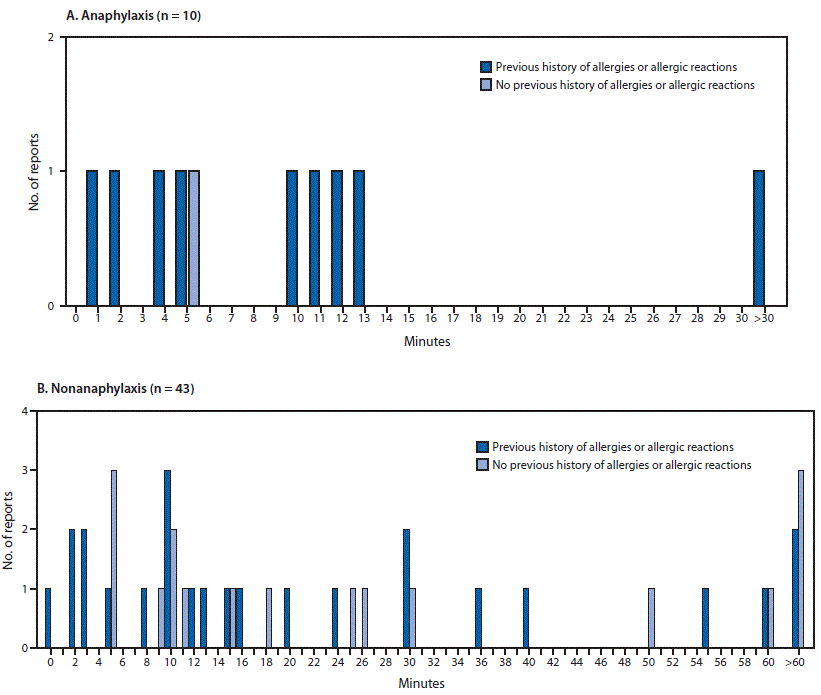
“Overall, severe allergic reactions to COVID-19 mRNA vaccines, including life-threatening anaphylaxis reactions such as low blood pressure and difficulty breathing, are rare, on the order of five cases per million vaccine doses administered,” noted Dr. Guerrerio, M.D., Ph.D., of NIAID’s Laboratory of Allergic Diseases, leads the trial. However, people who developed severe allergic reactions to a first dose of a COVID-19 mRNA vaccine are not eligible to enroll. The trial is seeking participants who experienced a mild or moderate systemic allergic reaction following a first dose of either the Pfizer-BioNTech or the Moderna COVID-19 mRNA vaccine. “This study will help us determine if individuals who experienced moderate systemic allergic reactions can safely receive a second dose of a COVID-19 mRNA vaccine.” “People who experienced an allergic reaction after receiving a COVID-19 mRNA vaccine may be hesitant to complete their vaccine regimen,” said Anthony S. Study participants will receive a second dose of vaccine as inpatients under carefully controlled conditions at the National Institutes of Health’s Clinical Center in Bethesda, Maryland. The single-site trial will enroll up to 100 people aged 16 to 69 years old who had an allergic reaction to a first dose of COVID-19 mRNA vaccines. Researchers from the National Institute of Allergy and Infectious Diseases (NIAID) are conducting a clinical trial designed to help understand rare but potentially serious systemic allergic reactions to COVID-19 mRNA vaccines. 
“The majority of these reactions developed within 15 minutes.Colorized scanning electron micrograph of chronically infected and partially lysed cells (blue) infected with a variant strain of SARS-CoV-2 virus particles (green), isolated from a patient sample. Robert Glatter, an emergency physician at Lenox Hill Hospital in New York. “In a recent study in JAMA, the incidence of anaphylaxis or severe allergic reactions after the Pfizer vaccine was rare, affecting just 11 people per million,” said Dr. In nine of them, onset occurred within 15 minutes of vaccination, with no anaphylaxis-related deaths reported. Kapusnik-Uner emphasized that true allergic reactions are rare, and may include rash or itching.Īccording to the Centers for Disease Control and Prevention (CDC), the vaccine adverse event reporting system (VAERS) detected 10 cases of anaphylaxis (severe allergic reaction) after administration of a little over 4 million first doses of the Moderna vaccine. “COVID-19 vaccines are administered with the intent of triggering the immune system and therefore can cause generalized symptoms for several days,” she said, “including fever, malaise, and fatigue while they are also working to mount their efficacious durable immunity via antibody production, and B cell & T cell responses.” Joan Kapusnik-Uner, PharmD, vice president of clinical content at First Databank (FDB), which publishes and maintains drug and medical device databases for healthcare professionals, said it’s important to know that there may be slight side effects from the vaccine that aren’t problematic. This vaccine is recommended for those 18 years and older, and is 94.1 percent effective. This vaccine is recommended for people 16 years and older, and is 95 percent effective at preventing illness. A Food and Drug Administration (FDA) emergency use authorization has been issued for two vaccines so far.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
